Exemption granted paves the way for early hospital implementation of aQuant.
MedTrace has received a positive response from the Danish Medicines Agency, granting exemption for the installation of its imaging software, aQuant, at two hospitals in Denmark – AUH and Region Hospital Gødstrup – ahead of final CE marking.
“This exemption marks a significant step forward for MedTrace, bringing us closer to making to a final aQuant widely available for clinical use,” says CEO Ann Kristin Led.
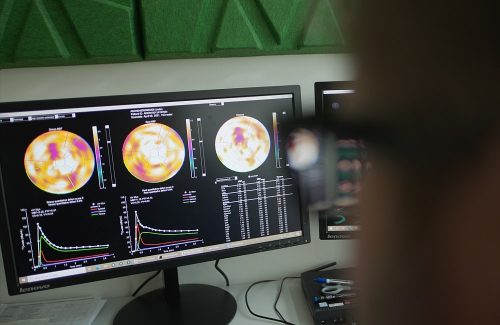
aQuant is a software suited for the semi-automated and robust analysis of 15O-water. The underlying algorithms of aQuant have been used in research and reported in more than 30 peer-reviewed articles over the last decade.
aQuant will, when cleared to market, offer analysis of the quantitative myocardial blood flow during PET exams with 15O-water.
// MedTrace’s technologies are not cleared to market for routine clinical use and are only available for use where an exemption has been granted by the relevant national authority.
Contact:t:
René Duvander
Director, Market Development
rene@medtrace.dk
About MedTrace
MedTrace is a pharma and medical device company with a 15O-water point-of-care solution under development. The solution consists of patented hardware for on-demand manufacturing, dosing, and infusion of 15O-water and patented software to analyze PET images. The company has an ongoing Phase III Clinical Trial, RAPID-WATER-FLOW, that seeks to evaluate the diagnostic accuracy and safety of 15O-water as a myocardial perfusion PET imaging agent.
We use technologies such as cookies to measure traffic and optimize the website. By clicking ‘Accept’ you consent to these technologies. Failure to consent may have a negative impact on certain features.